Radiation is all around us, from the natural background radiation on the earth to the man-made sources in our modern world. While radiation has many beneficial uses, such as medical imaging and cancer treatment, it can pose serious health risks if not properly controlled. In this article, we will explore the hidden hazards of radiation and how they can affect our health. From the dangers of radon in our homes to the risks of nuclear accidents, we will shine a light on the dark side of radiation and what we can do to protect ourselves.
History Of Radiation
Friends! Do you know who received the very first Nobel prize in Physics?

X-Ray: Very First Man-made Radiation
In 1895 the German physicist Wilhelm Conrad Röntgen, who was experimenting with cathode rays (streams of electrons) in a gas discharge tube observed that a screen coated with a fluorescent material placed near the tube would glow even when it was not directly in the path of the cathode rays.
He discovered that this new type of radiation could penetrate through many types of matter that were opaque to visible light and that it could produce images of the internal structure of solid objects, including human tissues. Röntgen called this new radiation “X-rays,”

When electrons moves at a tremendous speed from Cathode to Anode, out of 100 electrons one gives X-ray
Röntgen’s discovery revolutionized medical diagnosis and treatment, allowing doctors to see inside the human body without surgery. The first medical X-ray was taken by Röntgen himself of his wife’s hand, and the first medical use of X-rays was in diagnosing a broken bone.

Röntgen was awarded the Nobel Prize in Physics in 1901 for his discovery of X-rays, and the technology has continued to develop and improve over the past century, with applications in medicine, industry, and scientific research.
The era of WW-II, The Cold war & Nuclear Radiation
Radiation hazards have been recognized since the discovery of X-rays. However, it was not until the 1940s and 1950s that the full extent of radiation hazards became apparent.
During World War II, the United States government began to conduct extensive research into nuclear weapons, which required the development of methods to produce, store, and handle radioactive materials. This led to numerous accidents, such as the criticality accident at the Hanford Site in 1945, which resulted in the deaths of three workers and the release of a significant amount of radioactive material.

The most well-known incident involving radiation exposure occurred in 1986, with the explosion at the Chernobyl nuclear power plant in Ukraine. The explosion released a large amount of radioactive material into the environment, leading to numerous deaths and illnesses among workers and nearby residents.
Another well-known incident is the Fukushima nuclear disaster, which occurred in Japan in 2011. An earthquake and tsunami damaged the Fukushima Daiichi nuclear power plant, causing a release of radioactive material into the environment and resulting in the evacuation of nearby residents.
Which are Different Sources of Radiation?
Let us first know which are different sources of radiation.
It’s quite common to encounter radioactive materials in nature, which are typically not harmful in their natural state. Hence, it’s crucial to comprehend the proportion of naturally occurring radioactive material one encounter on a daily basis.
Natural Sources
1. Food: One of the most commonly encountered sources of natural radiation is one that many people don’t suspect. Bananas due to their high potassium content.

The Banana Equivalent Dose (BED) is an informal measure used to compare ionizing radiation exposure to the radiation dose one can receive from consuming an average-sized banana. It is intended as a simple educational tool to help people understand and contextualize levels of radioactivity.
2. Cosmic rays: the sun
A significant amount of cosmic radiation comes from sources beyond our solar system. Fortunately, Earth’s magnetosphere and the ozone layer intercept and absorb the majority of this energy, which is crucial for safeguarding life on our planet.
Individuals serving as flight crew on long-distance, high-altitude flights typically experience around 30% more annual radiation exposure compared to the average person.
3. Environment:
India is known to have high levels of terrestrial radiation in some regions due to the presence of radioactive minerals in the soil and rocks. The state of Kerala, located in the southern part of India, is known for its high levels of natural radiation. The region is enriched with monazite sand, which contains thorium, a radioactive element. As a result, people living in certain parts of Kerala are exposed to higher levels of radiation compared to other parts of India. However, the levels are generally within safe limits and do not pose a significant risk to human health.

4. Atmospheric: Radon, being a gas, can become a problem in some houses and other buildings, seeping in usually through cracks in solid foundations, and accumulating in rooms with poor ventilation.
Thoron is a radioactive gas that occurs naturally in the Earth’s atmosphere. However, the levels of Radon and thoron in the atmosphere are typically very low and do not pose a significant risk to human health.

5. Inside our body:
Yes It may surprise you but as our body is made up of elements present in our environment
‘यथा ब्रह्माण्डे तथा पिण्डे’
-चरक संहिता का यह श्लोकांश हमें समझाता है कि जो-जो इस ब्रह्माण्ड में है वही सब हमारे शरीर में भी है। यह भौतिक संसार पंचमहाभूतों से बना है- आकाश, वायु, अग्नि, जल और पृथ्वी। उसी प्रकार हमारा हमारा यह शरीर भी इन्हीं पाँचों महाभूतों से बना हुआ है।

Our body also contains radioactive materials like C-14 and potassium -40.
It is interesting to know that this decay of c-14 present in organisms is used to determine the age of the sample fairly accurately around 45000 years, known as radiocarbon dating!
All in all, it is estimated that the total natural radiation to which the average person is subjected comes to approximately 0.1 rad a year.
Man-made Sources of Radiation
1. X-ray:
Apart from natural sources of radiation, there are also human-made activities that generate radioactive materials that an average person might encounter frequently. However, the levels of exposure are relatively low.

Overall, the history of radiation hazards has been characterized by both the benefits and risks associated with the use of radiation. While radiation has many important applications, it is important to recognize and manage the potential hazards associated with its use.
Medical and dental X-rays currently represent the primary source of man-made radiation exposure to the general population.
X-rays have the potential to pose a health risk, particularly if someone is exposed to high doses over time. Patients, doctors, and medical technicians are at high risk.

When optimum radiographic techniques are employed, the skin dose to the patient from a single X-ray film varies roughly from 0.02 to 3.0 rad.
2. Nuclear fall-out
Nuclear power is perhaps the first thing that springs to mind when you ask someone about man-made sources of radiation.
A certain quantity of radioactive fallout was released into the atmosphere as a result of the detonation of atomic bombs in Hiroshima and Nagasaki, as well as nuclear weapons testing during the 1950s and 1960s.
Nuclear explosions release a tremendous amount of energy in the form of heat, light, ionizing radiation, and many radioactive. substances, the · important being the isotopes of carbon (C14), iodine (1 131), cesium (Cs 137), and strontium (Sr90). Cs137 and Sr90 are considered the most important because they are liberated in large amounts and remain radioactive for many years. The “half-life” of Sr90 is about 28 years and that of Cs137 is 30 years.

Global levels of atmospheric radioactive material were also affected by the 1986 explosion at the Chernobyl nuclear power plant in Ukraine, with a significantly greater impact in the immediate areas due to elements like Strontium-90 and Iodine-131. This resulted in a rise in thyroid cancer incidence.
3. Miscellaneous: Some everyday appliances (e.g., TV sets, and luminous wristwatches) are radioactive. But radiation from these sources at present is too small to be important.
Types of Radiation
Now let’s talk about types of radiation. Basically, it can be divided into ionizing and non-ionizing radiation by its ability to penetrate tissue or not respectively.
Ionizing Radiation
Radiation that is capable of penetrating tissues and depositing its energy within them is referred to as ionizing radiation.
There are two primary categories of ionizing radiation:
(1) electromagnetic radiations, such as X-rays and gamma rays, and
(2) corpuscular radiations, which include alpha particles, beta particles (electrons), and protons.
| Type Of Radiation | Approximate | Penetrating | Ability |
| Air | Tissues | Lead | |
| Alpha particles | 4 cm | .05 mm | 0 |
| Beta particles | 6-300 cm | 0.06-4.0 mm | 0.005-0.3mm |
| Gamma rays | 400 meter | 50cm | 40mm |
| X-rays | 120- 20 meter | 15-30 cm | 0.3mm |
| Cosmic rays | some components very high |
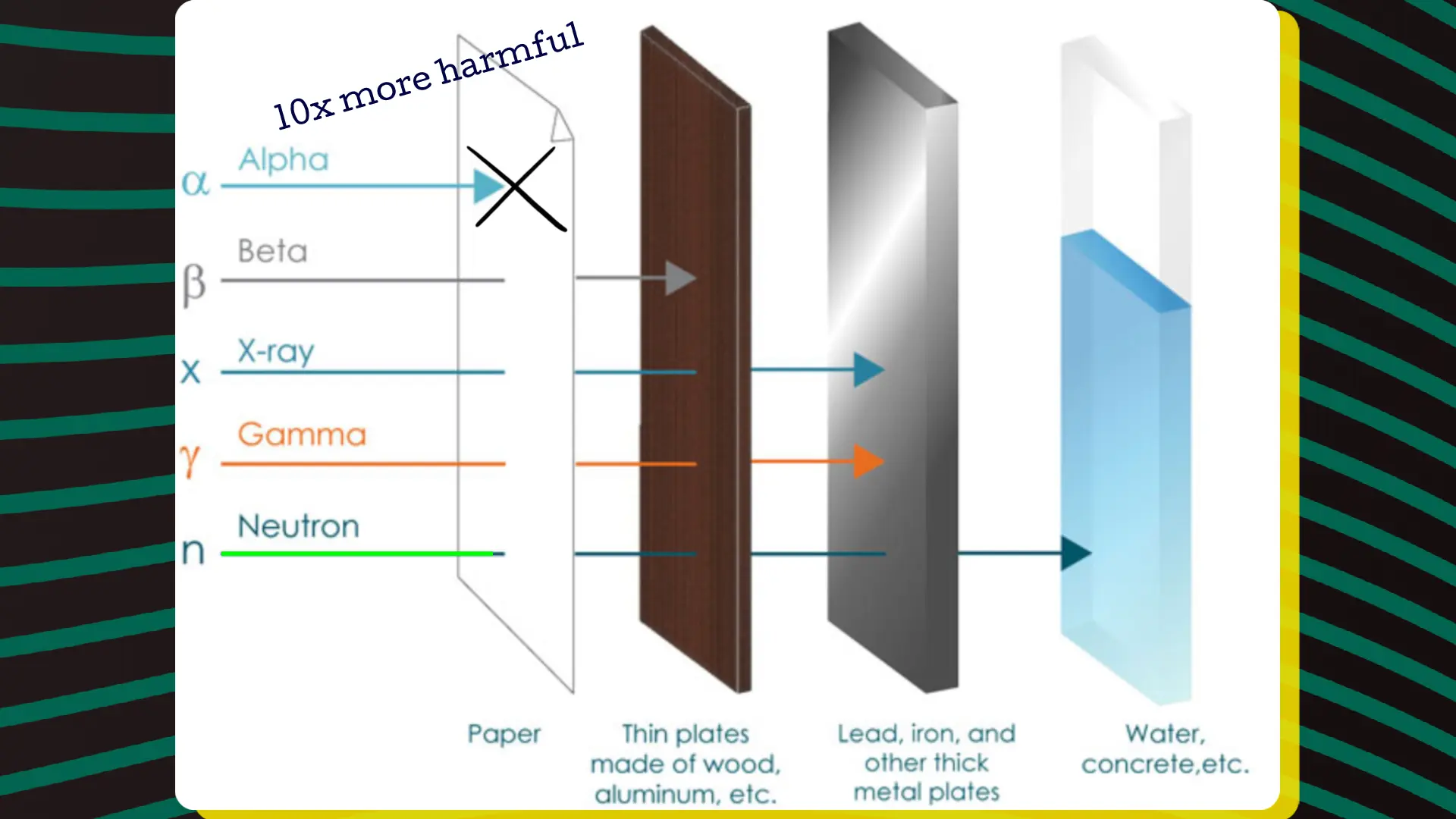
Compared to X-rays, beta particles, and gamma rays, alpha particles are 10 times more harmful. However, alpha particles do not have significant penetrating power, which is fortunate. Nevertheless, they pose a considerable risk if a radioactive substance has entered the body through inhalation or a wound. Gamma rays and X-rays, on the other hand, have short wavelengths and are highly penetrating. While X-rays are human-made, gamma rays occur spontaneously during the disintegration of radioactive elements. Other than that, there is no substantial difference between gamma rays and X-rays. Ionizing radiation is also present in cosmic rays.
Non-ionizing Radiation
Non-ionizing radiation refers to various forms of electromagnetic radiation that have wavelengths longer than those of ionizing radiation. As the wavelength increases, the energy of the electromagnetic radiation decreases. Therefore, all non-ionizing radiation has lower energy levels than cosmic, gamma, and X-ray radiation. Non-ionizing radiation is arranged in order of increasing wavelength, including ultraviolet (UV) radiation, visible light, infrared radiation, microwave radiation, and radio frequency radiation.
Biological Effects of Radiation

Radiation hazards are not limited to nuclear accidents, however. Many individuals have been exposed to radiation through medical procedures, such as X-rays and radiation therapy. While these procedures can be beneficial in treating and diagnosing medical conditions, they can also have harmful effects if not used properly.
Although it was Henri Becquerel that discovered the radiation phenomenon, it was his doctoral student, Marie Curie, who named it: radioactivity Curie died in 1934 of aplastic anaemia, probably developed from extended exposure to various radioactive materials, the dangers of which were only really understood long after most of her exposure had occurred. In fact, her papers (and even her cookbook) are still highly radioactive and many are considered unsafe to handle, stored in shielded boxes and requiring protective equipment to safely review.
RADIUM WATCH DIAL PAINTERS:

One of the first major events to highlight the dangers of ionizing radiation was the case of the “Radium Girls,” workers whose job was painting watch dials with radium.
Though there was enough suspicion of the effects of ionizing radiation for the management of the company to take precautions, they offered none to the actual workers painting the watch dials.
Many of them would lick their brushes to properly shape them. Since the human body treats radium as calcium, it was then deposited in the bones and led to radiation sickness. It is unknown how many died from radiation exposure.
THE MANHATTAN PROJECT & THE COLD WAR
The Manhattan Project, the crash study undertaken during World War II to develop the first atomic bomb, led directly to the second long-term study of the effects of long-term radiation exposure, namely the study of the survivors of the bombs at Hiroshima and Nagasaki. The bombings, which killed more than 150,000 between them (with some estimates putting the total at closer to 245,000 or more), also left more than 600,000 survivors (hibakusha, literally “explosion affected people”), many of who have been studied in the years since. Among the findings was that there does not appear to have been an increase in birth defects in those that survived the blasts. There have been, however, about 1900 cancer deaths that can be directly attributable to the bombings.
Biological effects of radiation are classified into two categories: acute or short-term effects resulting from exposure to high doses of radiation over a brief period, and chronic or long-term effects from exposure to low doses of radiation over an extended period.

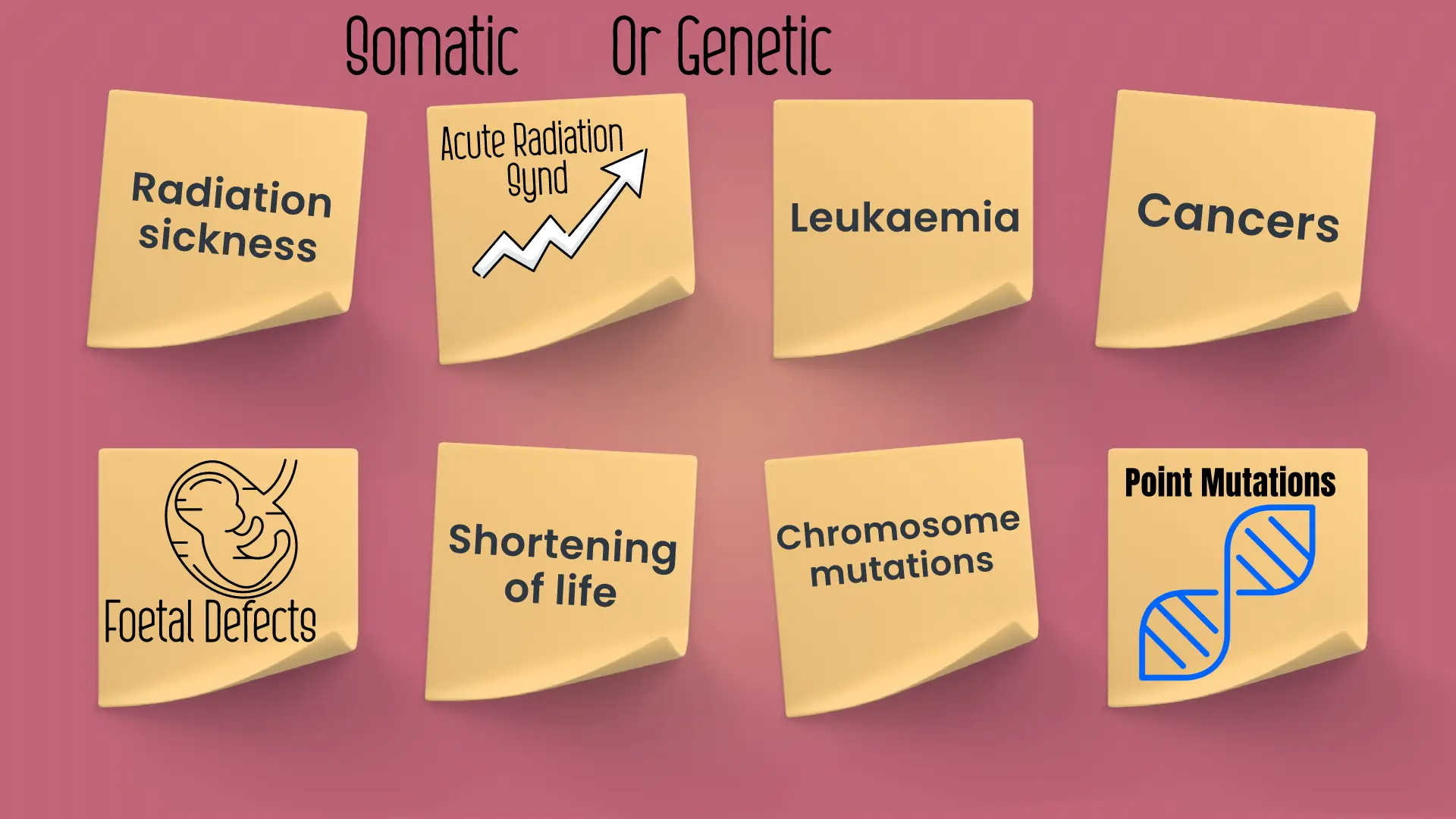
Somatic Effects:
High doses of radiation typically kill cells, leading to organ damage and possibly triggering an overall bodily response referred to as “Acute Radiation Syndrome”.
The effects of ionizing radiation can be somatic or genetic.
The biological response to the high dose of radiation is as per the dose
Like
< 5 rad: No immediate observable effects
– 5 rad to 50 rad: Slight blood changes may be detected by medical evaluations
– 50 rad to 150 rad: Slight blood changes will be noted and symptoms of nausea, fatigue, vomiting etc. likely.
– 150 rad to 1,100 rad: Severe blood changes will be noted and symptoms appear immediately. Approximately 2 weeks later, some of those exposed may die.
At about 300-500 rad, up to one-half of the people exposed will die within 60 days without intensive medical attention. Death is due to the destruction of the blood-forming organs. Without white blood cells, infection is likely. At the lower end of the dose range, isolation, antibiotics, and transfusions may provide the bone marrow time to generate new blood cells and full recovery is possible. At the upper end of the dose range, a bone marrow transplant may be required to produce new blood cells.
– 1, 100 rad to 2,000 rad: The probability of death increases to 100% within one to two weeks. The initial symptoms appear immediately. A few days later, things get very bad, very quickly since the gastrointestinal system is destroyed. Once the GI system ceases to function, nothing can be done, and medical care is for comfort only.
>2,000 rad: Death is a certainty. At doses above 5,000 rad, the central nervous system (brain and muscles) can no longer control body functions, including breathing and blood circulation. Everything happens very quickly. Nothing can be done, and medical care is for comfort only.
Genetic effect:
Somatic effects of radiation exposure are observable within the lifespan of the individual who has been irradiated, whereas genetic effects may be apparent in their distant offspring. Genetic effects arise from damage to chromosomes, including chromosome mutations and point mutations. Chromosome mutations may lead to sterility, while point mutations impact the genes directly.
How To Protect Ourselves From The Radiation Hazards?
Estimates suggest that the amount of radiation humans receive from outer space and background sources is around 0.1 rad per year, which is not currently considered a hazard. However, the allowable additional dose from human-made sources should not exceed 5 rad per year. Among human-made sources, X-rays are the most hazardous. Routine fluoroscopy, for instance, can deliver a dose of 4 rad to a specific body part in just a minute. Therefore, it is advisable to avoid unnecessary X-ray examinations, especially for children and pregnant women.
Regular House-keeping and Periodic Health Check-ups
Ensuring adequate control and surveillance of X-ray installations, protecting workers, improving techniques, and reducing doses are essential in preventing unnecessary exposure to radiation. Effective protective measures include using proper lead shields and lead rubber aprons. Lead aprons that contain 0.5mm of lead can reduce the intensity of scattered X-rays by over 90% and should be worn by all workers who regularly perform X-ray procedures. Workers must also wear a film badge or dosimeter that indicates their accumulated exposure to radiation since the last time the instrument was charged. Furthermore, workers’ health must be maintained by providing them with periodic medical examinations, regular working hours, recreational time, and holidays.
Radiation Hygiene
Radiation protection is a relatively new branch in the field of hygiene, known as radiation hygiene. Several organizations, including the International Commission on Radiological Protection (ICRP), the International Atomic Energy Agency (IAEA), and the World Health Organization (WHO), have been actively involved in this field.
It has been recommended that the genetic dose to the whole population from all sources addition to the natural background radiation, should not exceed 5 rems over a period of 30 years.
There has been a worldwide interest in preparing safety standards, and codes of practice for the safe operation of nuclear power plants and enunciating the basic principles of radiation protection.
How Radiation Is Measured?
International System of Units (SI)
| Unit | Name | Symbol |
| becquerel (Bq) | radioactive decay per second | Bq |
| gray (Gy) | the absorbed dose of ionizing radiation | Gy |
| sievert (Sv) | equivalent dose of ionizing radiation | Sv |
Traditional Units
| Unit | Name | Symbol |
| curie (Ci) | radioactivity of a substance | Ci |
| rad | radiation absorbed dose | rad |
| rem | equivalent dose of ionizing radiation (dose equivalent) | rem |
Commonly Used Conversion Factors
| To convert from | To | Multiply by |
| curie | becquerel | 3.7 x 10^10 |
| rad | gray | 0.01 |
| rem | sievert | 0.01 |
Note that the SI units are the preferred units for scientific and medical use, as they are internationally recognized and standardized. However, traditional units are still commonly used in some fields, particularly in the United States. It is important to be familiar with both systems of units when working with radiation.
FAQs On Radiation
What are ionizing radiations?
Ionizing radiation refers to any type of radiation that has enough energy to ionize atoms and molecules, which means it can strip electrons from them, resulting in charged particles (ions). Examples of ionizing radiation include X-rays, gamma rays, and alpha and beta particles.
How are ionizing radiations produced?
Ionizing radiation can be produced naturally or artificially. Natural sources include cosmic rays, radon gas, and radioactive materials in the earth’s crust. Artificial sources include X-ray machines, nuclear power plants, and nuclear weapons.
What are the health effects of ionizing radiation?
Ionizing radiation can cause damage to living cells and tissues, which can result in acute and long-term health effects. Acute effects may include radiation sickness, burns, and damage to the immune system. Long-term effects may include an increased risk of cancer and genetic mutations.
What are the units used to measure ionizing radiation?
The units used to measure ionizing radiation include the Gray (Gy) and the Sievert (Sv). The Gray measures the amount of energy absorbed by a material from ionizing radiation, while the Sievert measures the effective dose of radiation that is received by living tissue.
How can people protect themselves from ionizing radiation?
People can protect themselves from ionizing radiation by limiting exposure to sources of radiation, using protective equipment such as lead aprons and shields, and following safety protocols when working with radioactive materials.
What are some of the regulations governing ionizing radiation in India?
In India, the Atomic Energy Regulatory Board (AERB) is responsible for regulating ionizing radiation. Some of the regulations governing ionizing radiation in India include:
- Atomic Energy Act (1962): This act provides for the development, control, and use of atomic energy for peaceful purposes and for the regulation of research, production, and use of radioactive materials.
- Atomic Energy (Radiation Protection) Rules (2004): These rules provide for the protection of workers and the public from ionizing radiation by setting exposure limits, establishing safety procedures, and requiring monitoring and record keeping.
- Radiation Safety Standards (2011): These standards provide guidance on the safe use of radiation sources and equipment, including requirements for registration and licensing, inspection and enforcement, and emergency preparedness and response.
- National Disaster Management Guidelines on Nuclear and Radiological Emergencies (2019): These guidelines provide a framework for managing nuclear and radiological emergencies, including emergency response procedures, communication protocols, and coordination mechanisms.
- Bureau of Indian Standards (BIS): BIS sets standards for radiation measurement instruments, protective equipment, and procedures to ensure that they meet the required safety standards.
What are non-ionizing radiations?
Non-ionizing radiation refers to any type of radiation that does not have enough energy to ionize atoms and molecules. Examples of non-ionizing radiation include radio waves, microwaves, infrared radiation, visible light, and ultraviolet radiation.
How are non-ionizing radiations produced?
Non-ionizing radiation can be produced naturally or artificially. Natural sources include sunlight, the earth’s magnetic field, and lightning. Artificial sources include radio and television broadcasts, microwave ovens, and certain medical devices.
What are the health effects of non-ionizing radiation?
Non-ionizing radiation can cause both acute and long-term health effects, depending on the intensity and duration of exposure. Acute effects may include skin burns and eye damage from exposure to ultraviolet radiation, while long-term effects may include an increased risk of skin cancer and cataracts.
What are the units used to measure non-ionizing radiation?
The units used to measure non-ionizing radiation depends on the specific type of radiation. For example, radio waves are measured in hertz (Hz), microwaves are measured in gigahertz (GHz), and ultraviolet radiation is measured in nanometers (nm).
How can people protect themselves from non-ionizing radiation?
People can protect themselves from non-ionizing radiation by limiting exposure to sources of radiation, such as by using protective equipment like sunglasses and sunscreen when exposed to sunlight, and following safety protocols when working with equipment that emits non-ionizing radiation.

Add a Comment
You must be logged in to post a comment